For a while the big conversations around the environment and climate were climate change, pollution and the ozone hole. The climate is most definitely still changing, and pollution especially in the form of plastics is still a popular point of discussion, but what about ozone? What is it, and why was it a hole? Why was this bad? And is it still a hole? Basically, what’s going on with ozone? Well luckily your questions will be answered in this week’s plog! Enjoy!
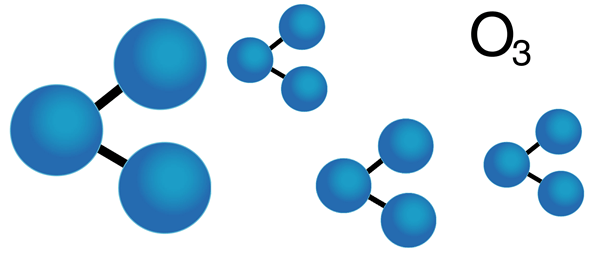
‘Good’ Ozone
The air we breathe is made up of 21% oxygen, a colourless gas made up of sets of two oxygen atoms joined together (O2). However, oxygen can also exist as a colourless gas made up of three oxygen atoms (O3), which we call ozone. Ozone can be found high up in the atmosphere as well as nearer to the ground, and depending on where it is it can either be great, or really quite problematic.
The ozone high up in the atmosphere, called stratospheric ozone as it is in the bottom of the stratosphere, is good. This sits around 15-30 km above the earth’s surface where it has an important protective role absorbing ultraviolet (UV) radiation from the sun. We need some UV light. This light, which is in a colour spectrum beyond our ability to see, allows us to make vitamin D when we are exposed to it, it stimulates the production of feel-good hormones in the brain and it can help certain birds, reptiles and insects navigate as they see in a slightly different colour spectrum from us. UV can also be a great tool for sterilising as it can deactivate or destroy some microbes by destroying their DNA.
However… if UV has the power to mutate and destroy microbes… that hints it’s not all good for us. Too much UV can cause skin cancer, sunburn (which can lead to skin cancer), suppress the immune system and damage our eyes. This is why we need only a middling amount of UV, enough to get the good but not so much that we get the bad. The ozone in the atmosphere absorbs much of the UV that comes through the helping us to keep this moderate balance. So stratospheric ozone is good and we need it, but what about ground level ozone, also known as ‘tropospheric ozone’?

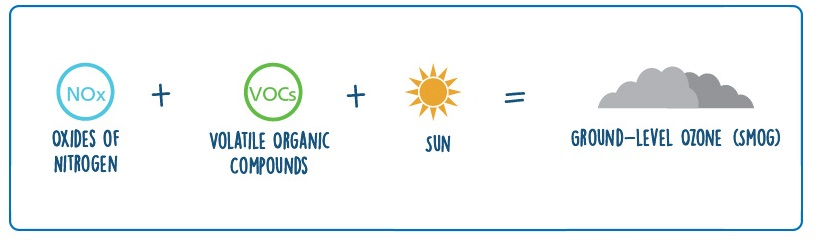
‘Bad’ Ozone
Ground level ozone is often created out of a mix of nitrous oxide, and volatile organic compounds reacting together, these being produced by e.g. cars, power plants, refineries, chemical plants etc… and can be carried by the wind far away from these areas of production. Ozone production is highest on warm, sunny days.
When ozone is breathed in it can cause muscles in our airways to tighten making breathing more difficult, causing inflammation and damage to the airways, aggravating pre-existing lung conditions like asthma and bronchitis and making the lungs more susceptible to infection. All round not-ideal. Dealing with this involves having tighter regulation on the filtering of gases emitted by industrial processes and cars, as well as just producing less of these harmful by-products in the first place e.g. by lowing individual car use and increasing public transport or cycling infrastructure where possible.
So whilst we want lots of ozone high up to absorb UV radiation, it is definitely something we want to avoid on the ground due to it’s health impacts.

The Ozone Hole
So, the bad ground ozone fluctuates depending on the heat, sunshine, and how much nitrous oxide and volatile organic compounds are being produced. What about the good stratospheric ozone?
Stratospheric ozone normally fluctuates throughout the year due to changes in temperature, season and the action of winds such as the polar vortex moving air around even high up in the atmosphere. However, in the 1970s some chemists at the University of California published a scientific paper pointing out a bit of an issue. Chlorofluorocarbon gases (CFCs) when reaching the stratosphere were broken down by UV light into a variety of substances including chlorine which destroys ozone. In fact one chorine atom had the potential to destroy over 100,000 ozone molecules.
This problem was made all the more problematic by the fact the CFCs were in loads of things. Fridges in every home contained CFCs, as well as aerosols, meaning every time someone sprayed on some hairspray or deodorant CFCs were released, chlorine produced and ozone destroyed. All this, it was noticed, led to the creation of the famous ozone hole.
So this ‘hole’ wasn’t actually a hole, but a region of low ozone concentration over Antarctica. It also wasn’t constantly there- with all the seasonal fluctuations it was only around between August and October. However, it was a problem, as with less ozone periodically in the area, more UV light was entering the earth’s atmosphere and so increasing the risk of burns, mutations and cancers. Whilst this one big Antarctic ‘hole’ was probably the most famous, other parts of the ozone layer were thinning out too all over the world.
If you’re ever feeling like environmental change and saving is hopeless and futile, here is a wonderful saving example. Recognising the massive issues to the ozone layer caused by CFCs and the subsequent harmful impacts on health, the Montreal Protocol was written up which led to the phasing out of ozone depleting substances across all UN countries, that’s 197 in total. This reduced by millions the potential number of skin cancers and cataracts that could have occurred as well as slowing down the warming of the planet. It took some time for this to have an impact of course, as even though CFC’s weren’t being newly released there were many still lingering. However after 30 years the ozone hole started to recover. It is thought that the holes in the northern hemisphere will be fully recovered by the 2030s, the ones in the southern hemisphere by 2050s and those over the polar regions by the 2060s. They’re already well on their way to recovery!!! Of course it’s not perfect, CFCs still are used in some places, and some of the things we’ve replaced CFCs with aren’t great for ozone either, but it is still huge progress!
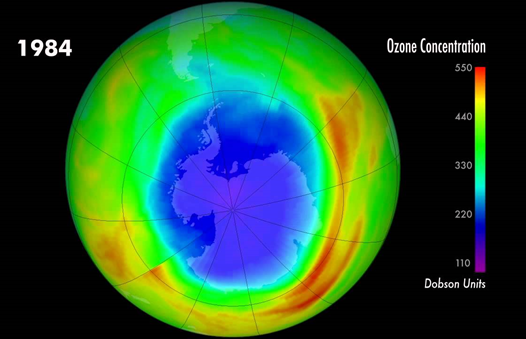
So in a vein that we’re not really used to in the environmental world, good climate things really are happening on this front. Good stratospheric ozone is on the increase, filling in those seasonal ozone ‘holes’, and whilst the ‘bad’ tropospheric or ground level ozone hasn’t been so strictly regulated, the recent mass reduction in driving due to the lockdown has been widely noted to create improved air quality, part of which is reduced ozone. Perhaps with so many being finally able to breathe fresh air in cities again, we may want to keep this feature for after the lockdown, and reduce the ground ozone for good. Fingers crossed!
For more info:
A scientific paper about the yearly fluctuations in ozone: https://earth-planets-space.springeropen.com/articles/10.1186/s40623-017-0774-4
National Geographic article on ozone depletion: https://www.nationalgeographic.com/environment/global-warming/ozone-depletion/
Music: kongano.com


Leave a comment